
Training Medical Device Single Audit Program Mdsap The medical device single audit program, or mdsap for short, is a regulatory framework that allows medical device manufacturers to undergo a single audit of their quality management system (qms) that satisfies the requirements of multiple regulatory authorities simultaneously. Our vision for the mdsap is to ensure the highest standards of safety for medical devices, whilst minimizing regulatory burden through use of an agreed single audit process.

Mdsap Archives Learngxp Accredited Online Life Science Training Courses This page contains information about the medical device single audit program (mdsap). Mdsap is a global auditing initiative that allows recognized third party auditors to conduct a single regulatory audit of a medical device manufacturer. this audit is designed to meet the qms requirements of multiple participating regulatory authorities. The objective of the mdsap is to develop, manage, and oversee a single audit program that will allow a single regulatory audit of a medical device manufacturer, conducted by an mdsap recognised auditing organisation, to satisfy the needs of multiple regulatory jurisdictions. The medical device single audit program, or mdsap for short, is a regulatory framework that allows medical device manufacturers to undergo a single audit of their quality management system (qms).

Mdsap Faq The objective of the mdsap is to develop, manage, and oversee a single audit program that will allow a single regulatory audit of a medical device manufacturer, conducted by an mdsap recognised auditing organisation, to satisfy the needs of multiple regulatory jurisdictions. The medical device single audit program, or mdsap for short, is a regulatory framework that allows medical device manufacturers to undergo a single audit of their quality management system (qms). The medical device single audit program (mdsap) allows a single audit of a medical device manufacturer’s quality management system (qms), which satisfies the requirements of multiple regulatory jurisdictions. Learn the basics of the medical device single audit program (mdsa) and mdsap auditing in 2023. In this guide, we’ll explore what mdsap is, its benefits, the necessary requirements, and how to avoid common mistakes that may hinder the certification process. The mdsap streamlines regulatory compliance by allowing a single audit to satisfy multiple countries' requirements, reducing the need for duplicative inspections. it enhances global market access and predictability for manufacturers while promoting consistent quality system oversight.

Mdsap Simplifying Compliance For Medical Device Manufacturers The medical device single audit program (mdsap) allows a single audit of a medical device manufacturer’s quality management system (qms), which satisfies the requirements of multiple regulatory jurisdictions. Learn the basics of the medical device single audit program (mdsa) and mdsap auditing in 2023. In this guide, we’ll explore what mdsap is, its benefits, the necessary requirements, and how to avoid common mistakes that may hinder the certification process. The mdsap streamlines regulatory compliance by allowing a single audit to satisfy multiple countries' requirements, reducing the need for duplicative inspections. it enhances global market access and predictability for manufacturers while promoting consistent quality system oversight.
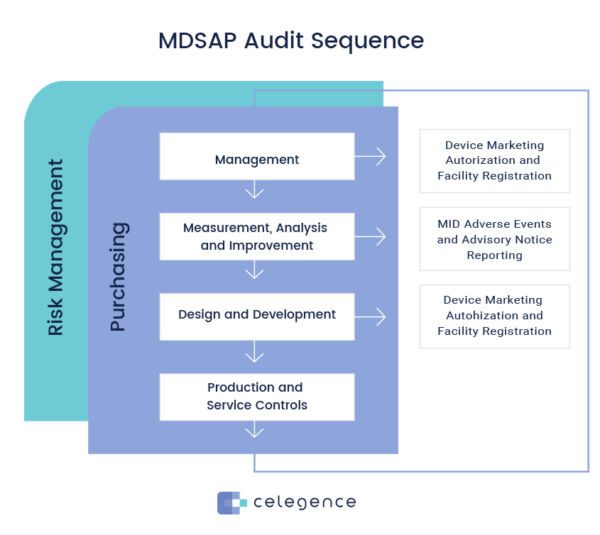
What Is The Mdsap Audit Approach Guide To Mdsap Celegence In this guide, we’ll explore what mdsap is, its benefits, the necessary requirements, and how to avoid common mistakes that may hinder the certification process. The mdsap streamlines regulatory compliance by allowing a single audit to satisfy multiple countries' requirements, reducing the need for duplicative inspections. it enhances global market access and predictability for manufacturers while promoting consistent quality system oversight.

Mdsap Pptx