
Atomic Mass And Atomic Number Pdf Atoms Chemical Elements Elements consist of atoms. every atom of an element has the same number of protons. this number is referred to as the atomic number. atoms of an element may have a varying number of neutrons. the number of protons plus the number of neutrons is known as the mass number. The mass number (represented by the letter a) is defined as the total number of protons and neutrons in an atom. consider the table below, which shows data from the first six elements of the periodic table.

Understanding Atomic Number And Atomic Mass Instructional Video For 9th Here’s the reality: atomic mass and atomic number are completely different concepts that serve distinct purposes in chemistry. one determines what element you’re looking at, while the other tells you how heavy it is. confuse these two, and your stoichiometry calculations will be completely wrong. Discover the differences between atomic mass and atomic number, including their definitions, calculations, and applications in chemistry and beyond. learn how these fundamental concepts impact our understanding of elements properties and behavior. Atomic number tells us about the number of electrons or protons, whereas atomic mass tells us about the number of neutrons and protons combined. these two parameters are important in learning about atoms and their various properties. Atomic number is the number of protons in an atom of an element. it is a whole number or the symbol z in general notation. it isn’t always listed because you can figure out the number of protons if you know the element symbol. mass number is the sum of the number of protons and neutrons in an atom.
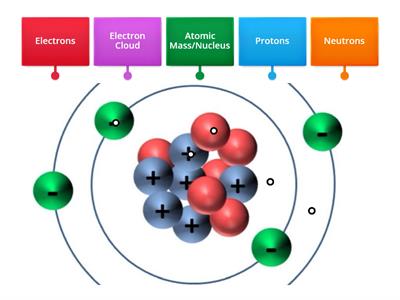
Atomic Number And Atomic Mass Teaching Resources Atomic number tells us about the number of electrons or protons, whereas atomic mass tells us about the number of neutrons and protons combined. these two parameters are important in learning about atoms and their various properties. Atomic number is the number of protons in an atom of an element. it is a whole number or the symbol z in general notation. it isn’t always listed because you can figure out the number of protons if you know the element symbol. mass number is the sum of the number of protons and neutrons in an atom. Therefore, the atomic number tells you what element an atom is, while the atomic mass provides information about how heavy an average atom of that element is, accounting for its various isotopic forms. In chemistry the amount of protons in an atom is referred to as its atomic number. this is usually denoted by the letter “z.” an atom is usually made up of protons neutrons and electrons, but since electrons are generally so small only protons and neutrons account for the mass of an element. The sum of relative isotopic masses of all atoms in a molecule is the relative molecular mass. the atomic mass of an isotope and the relative isotopic mass refers to a certain specific isotope of an element. Atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons. in fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); the number of protons in an atom is called the atomic number.

Mass Number Versus Atomic Number And Atomic Mass Therefore, the atomic number tells you what element an atom is, while the atomic mass provides information about how heavy an average atom of that element is, accounting for its various isotopic forms. In chemistry the amount of protons in an atom is referred to as its atomic number. this is usually denoted by the letter “z.” an atom is usually made up of protons neutrons and electrons, but since electrons are generally so small only protons and neutrons account for the mass of an element. The sum of relative isotopic masses of all atoms in a molecule is the relative molecular mass. the atomic mass of an isotope and the relative isotopic mass refers to a certain specific isotope of an element. Atomic number, atomic mass, and relative atomic mass atoms of each element contain a characteristic number of protons. in fact, the number of protons determines what atom we are looking at (e.g., all atoms with six protons are carbon atoms); the number of protons in an atom is called the atomic number.