
Lanthanides And Actinides Pdf The document outlines the course chm 315: inorganic chemistry iii (part b), focusing on the f block transition elements, specifically lanthanides and actinides. it covers their discovery, occurrence, extraction methods, electronic structure, general properties, and oxidation states. The russel saunders coupling scheme is used for the lighter metals and the j j coupling scheme is used for heavier atoms such as lanthanides and actinides. to get the term symbol for the atom or ion in the ground state, it is designated as s, p, d or f depending on the value of l.

Chemistry Of Lanthanides Pdf Lanthanide Coordination Complex Electronic configurations of actinides are not always easy to confirm. atomic spectra of heavy elements are very difficult to interpret in terms of configuration. To bring the student to the understanding of the nature of lanthanide and actinide contractions, as it affects the properties of the metals. to explain the reasons for variable oxidation states of the metals and the reactivity of the respective oxidation state. As radioactive lanthanide isotopes are important fission products of the fission of 235u and therefore need to be separated from uranium, and be cause the actinides after plutonium tend to resemble the lanthanides, the development of the technique followed on the manhattan project. Definition: the f block (inner transition) elements containing partially filled 4f subshells are known as lanthanides or lanthanones because of their close similarities with element lanthanum (atomic no: 57). the fourteen elements from atomic no: 58 to 71 constitute lanthanides.

The Lanthanides And Their Complexes Pdf Lanthanide Physical Sciences As radioactive lanthanide isotopes are important fission products of the fission of 235u and therefore need to be separated from uranium, and be cause the actinides after plutonium tend to resemble the lanthanides, the development of the technique followed on the manhattan project. Definition: the f block (inner transition) elements containing partially filled 4f subshells are known as lanthanides or lanthanones because of their close similarities with element lanthanum (atomic no: 57). the fourteen elements from atomic no: 58 to 71 constitute lanthanides. (16) reducing properties : transition metals having negative standard reduction potential should be good reducing agents i.e. they should be oxidised easily to their ions. Book available to patrons with print disabilities. Generic descriptions of the phase behaviors in the lanthanide and actinide hydrides, as well as a thorough examination and explanation of the thermodynamic relationships of these metal hydrogen systems, are given by john w. ward and john m. haschke in chapter 123. Pdf | this review summarizes the progress in organo f element chemistry during the year 2018.

The Transition Metals The Lanthanides And The Antinides (16) reducing properties : transition metals having negative standard reduction potential should be good reducing agents i.e. they should be oxidised easily to their ions. Book available to patrons with print disabilities. Generic descriptions of the phase behaviors in the lanthanide and actinide hydrides, as well as a thorough examination and explanation of the thermodynamic relationships of these metal hydrogen systems, are given by john w. ward and john m. haschke in chapter 123. Pdf | this review summarizes the progress in organo f element chemistry during the year 2018.
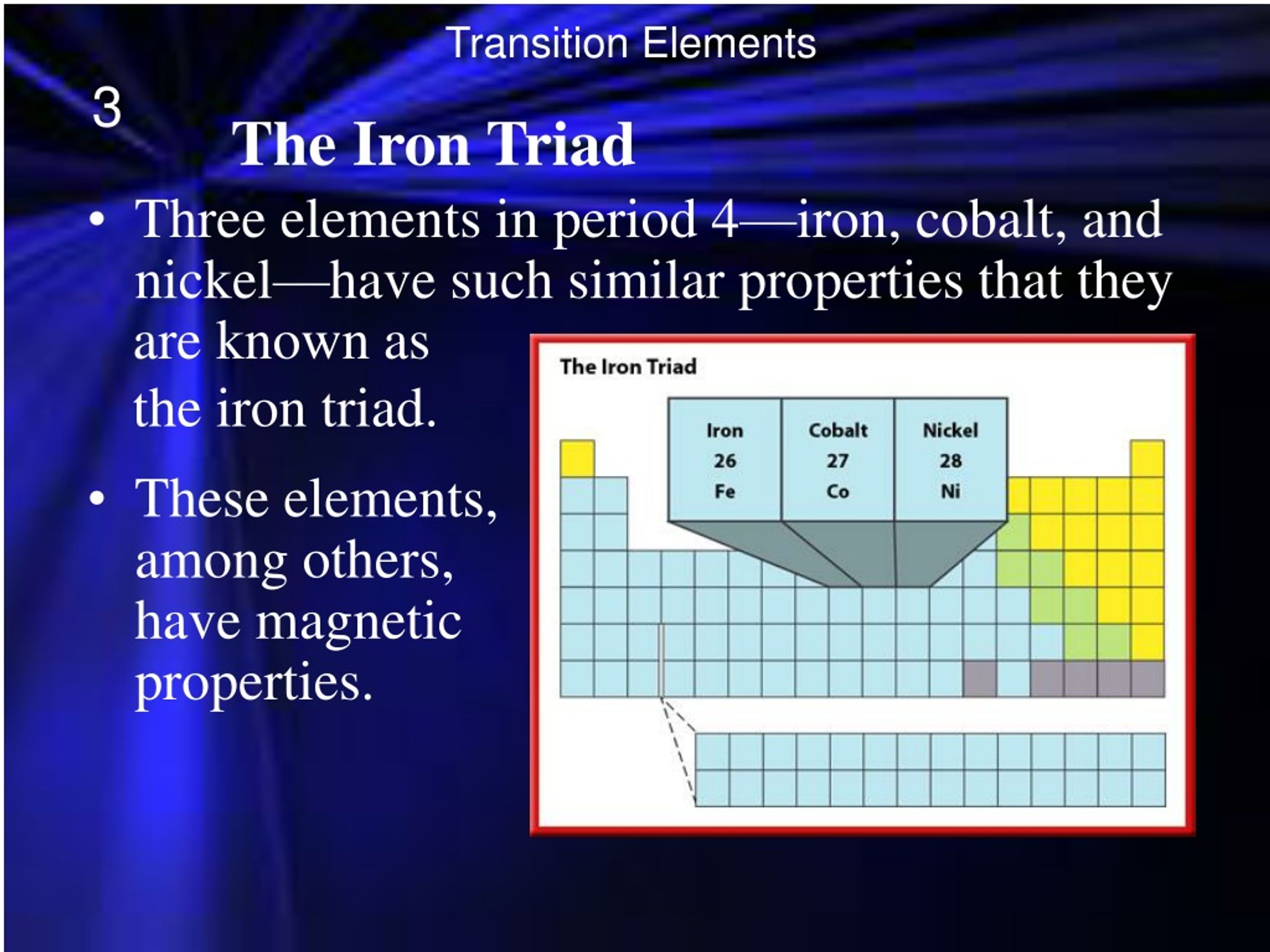
Ppt The Transition Metals The Lanthanides And The Antinides Generic descriptions of the phase behaviors in the lanthanide and actinide hydrides, as well as a thorough examination and explanation of the thermodynamic relationships of these metal hydrogen systems, are given by john w. ward and john m. haschke in chapter 123. Pdf | this review summarizes the progress in organo f element chemistry during the year 2018.

Ppt The Transition Metals The Lanthanides And The Antinides