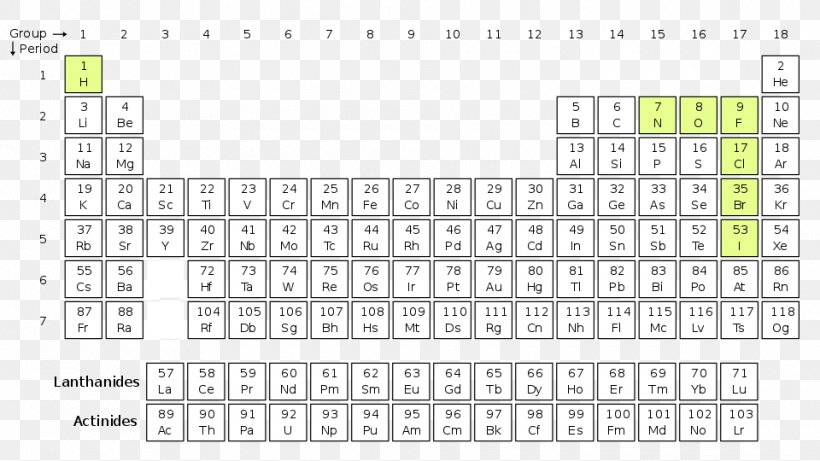
Lanthanides And Actinides Periodic Table Lanthanides And Actinides This page discusses russian nesting dolls as a metaphor for the layered structure of lanthanides and actinides in the periodic table. lanthanides (atomic numbers 58 71) are used in alloys and optical …. Lanthanides and actinides are separate because of their unique electron configurations and f block properties. both groups belong to the 3b elements, which are the first to start filling d shell electrons.
Lanthanides And Actinides Periodic Table Lanthanides And Actinides Lanthanides and actinides are two prominent families of the periodic table of elements. discussed below are the differences between the elements in these two families and their common properties. The lanthanides are located on the top row of the two rows of elements found below the main body of the periodic table. placing the lanthanides and the actinides (the row below the lanthanides) below the other elements is just a matter of aesthetics. The lanthanides and the actinides make up the f block of the periodic table. the lanthanides are the elements produced as the 4 f sublevel is filled with electrons and the actinides are formed while filling the 5 f sublevel. Lanthanides and actinides are two separate series of metallic elements found at the bottom of the periodic table. they are collectively known as inner transition elements because their differentiating electron enters the (n 2)f orbital, which is an inner orbital compared to the outermost shell.

Lanthanides And Actinides Periodic Table Actinides Mendeleev Table The lanthanides and the actinides make up the f block of the periodic table. the lanthanides are the elements produced as the 4 f sublevel is filled with electrons and the actinides are formed while filling the 5 f sublevel. Lanthanides and actinides are two separate series of metallic elements found at the bottom of the periodic table. they are collectively known as inner transition elements because their differentiating electron enters the (n 2)f orbital, which is an inner orbital compared to the outermost shell. Lanthanides and actinides differ from other elements in the periodic table due to their unique electronic configuration. they exhibit different chemical properties compared to the transition metals and the main group elements. This guide breaks down what lanthanides and actinides actually are. you will see why chemists organized them this way and how to work with them confidently during tests. The document provides an overview of lanthanides and actinides, detailing their definitions, properties, similarities, differences, applications, and interesting facts. Lanthanides and actinides play a crucial role in modern technology, medicine, energy, and research. their unique properties make them valuable in various applications.
/141849999-56a12e8b3df78cf77268332b.jpg)
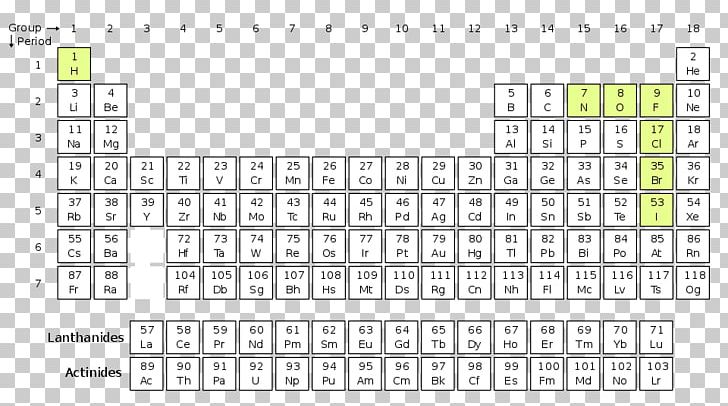
Why Lanthanides And Actinides Are Separate On The Periodic Table Lanthanides and actinides differ from other elements in the periodic table due to their unique electronic configuration. they exhibit different chemical properties compared to the transition metals and the main group elements. This guide breaks down what lanthanides and actinides actually are. you will see why chemists organized them this way and how to work with them confidently during tests. The document provides an overview of lanthanides and actinides, detailing their definitions, properties, similarities, differences, applications, and interesting facts. Lanthanides and actinides play a crucial role in modern technology, medicine, energy, and research. their unique properties make them valuable in various applications.