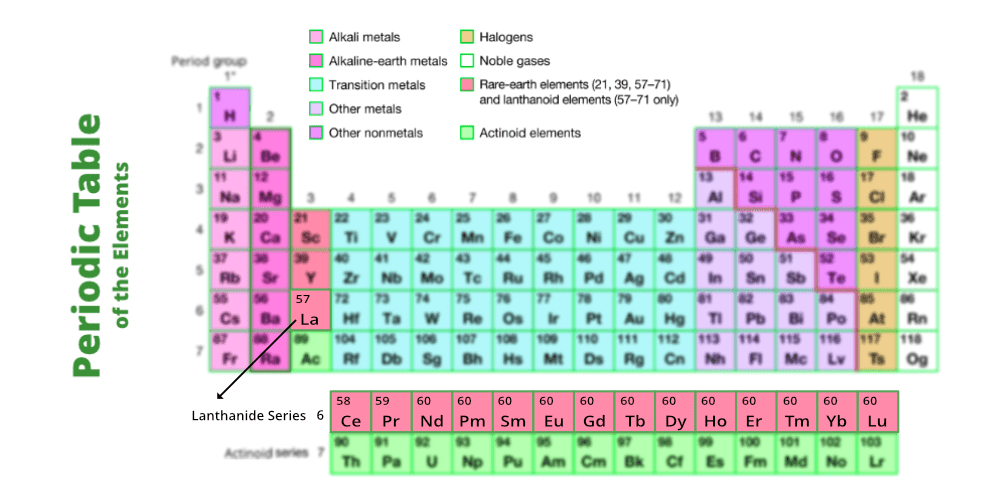
Lanthanide Elements All lanthanide elements form trivalent cations, ln 3 , whose chemistry is largely determined by the ionic radius, which decreases steadily from lanthanum (la) to lutetium (lu). Chemists refer to these 15 elements using the symbol ln. they are called “lanthanides” because they share properties in common with lanthanum. here is a list of the lanthanides, a look at their common properties, their uses, and other facts.
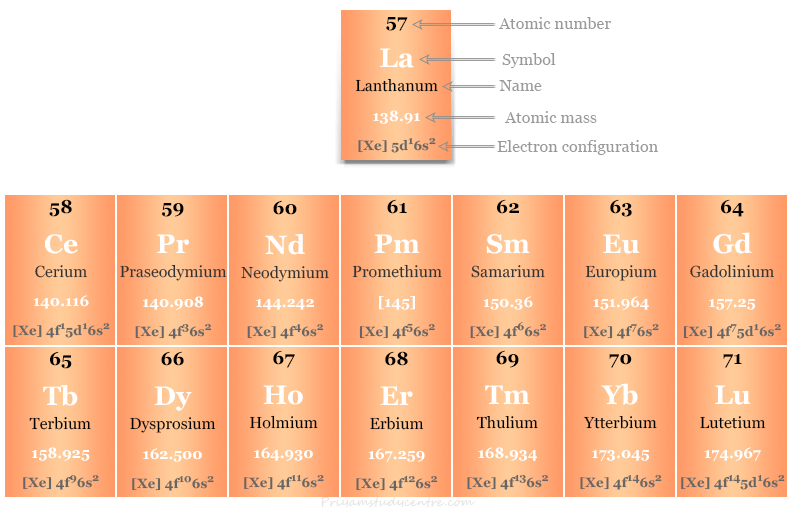
Lanthanide Elements Lanthanides specifically refer to the metallic elements in the lanthanide series of the periodic table, from lanthanum to lutetium. heavy metals are distributed across various groups on the periodic table, primarily in the transition metals and post transition metals. Lanthanide, any of the series of 15 consecutive chemical elements in the periodic table from lanthanum to lutetium (atomic numbers 57–71). with scandium and yttrium, they make up the rare earth metals. Here's a list of elements that are lanthanides, a subset of the rare earth group of elements. also learn about lanthanide properties. The lanthanides consist of the elements in the f block of period six in the periodic table. while these metals can be considered transition metals, they have properties that set them apart from the rest of the elements.

Lanthanide Elements Here's a list of elements that are lanthanides, a subset of the rare earth group of elements. also learn about lanthanide properties. The lanthanides consist of the elements in the f block of period six in the periodic table. while these metals can be considered transition metals, they have properties that set them apart from the rest of the elements. What are the lanthanides, aka rare earth elements? the lanthanide series is a row of elements situated below the main block of the periodic table, within the f block along with the actinides. they are all metals. lanthanides occupy elements 57 71 and branch off after barium on the periodic table. Lanthanides are a set of 15 chemical elements whose atomic numbers range from 57 to 71 with one valence electron in the 5d shell. The lanthanides refer to lanthanum and 14 other elements that are chemically similar to lanthanum. lanthanides find the most applications in catalysts and glass production. but from a value standpoint, their use in phosphors and magnets is more important. The lanthanide series refers to the 14 elements that result from the addition of electrons into the 4f orbitals, characterized primarily by the formation of trivalent m³⁺ ions and notable for their ionic compounds with coordination numbers typically of 8 or 9.
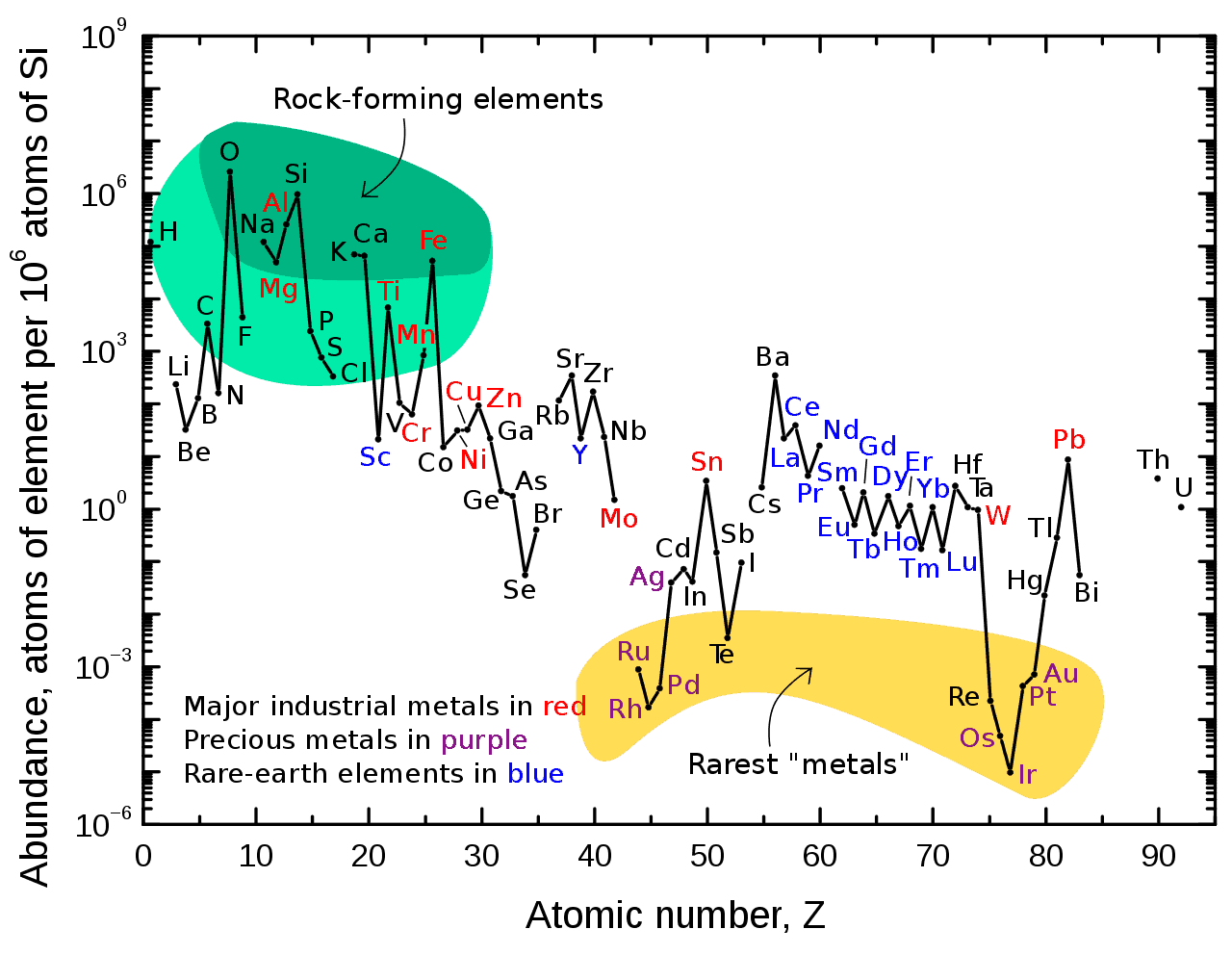
Lanthanide Elements What are the lanthanides, aka rare earth elements? the lanthanide series is a row of elements situated below the main block of the periodic table, within the f block along with the actinides. they are all metals. lanthanides occupy elements 57 71 and branch off after barium on the periodic table. Lanthanides are a set of 15 chemical elements whose atomic numbers range from 57 to 71 with one valence electron in the 5d shell. The lanthanides refer to lanthanum and 14 other elements that are chemically similar to lanthanum. lanthanides find the most applications in catalysts and glass production. but from a value standpoint, their use in phosphors and magnets is more important. The lanthanide series refers to the 14 elements that result from the addition of electrons into the 4f orbitals, characterized primarily by the formation of trivalent m³⁺ ions and notable for their ionic compounds with coordination numbers typically of 8 or 9.

Lanthanide Elements The lanthanides refer to lanthanum and 14 other elements that are chemically similar to lanthanum. lanthanides find the most applications in catalysts and glass production. but from a value standpoint, their use in phosphors and magnets is more important. The lanthanide series refers to the 14 elements that result from the addition of electrons into the 4f orbitals, characterized primarily by the formation of trivalent m³⁺ ions and notable for their ionic compounds with coordination numbers typically of 8 or 9.