
Isotope Release 4 Funky New Limited Edition Hydriums The term isotope comes from the greek roots isos (ἴσος "equal") and topos (τόπος "place"), meaning "the same place": different isotopes of an element occupy the same place on the periodic table. [2]. An isotope is one of two or more species of atoms of a chemical element with the same atomic number and position in the periodic table and nearly identical chemical behavior but with different atomic masses and physical properties.
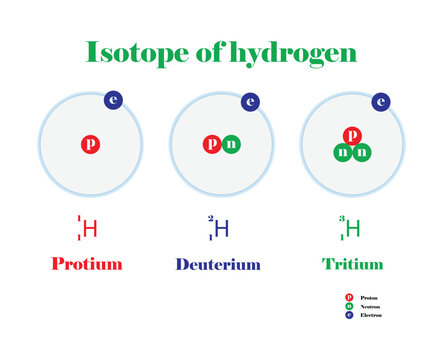
ร ปภาพisotope เล อกด ภาพถ ายสต อก เวกเตอร และว ด โอ8 246 Adobe Stock Learn what an isotope is, how to write it, and how to distinguish it from a nuclide. find out the difference between stable and radioactive isotopes, and see examples of isotopes of hydrogen, carbon, and uranium. Identify the most abundant isotope when given specific values. calculate the atomic mass of an element from the masses and relative percentages of the isotopes of the element. Carbon 12 is a stable isotope, while carbon 14 is a radioactive isotope (radioisotope). other than this, many synthetic carbon isotopes are synthesized in a lab environment and can only be sustained for a very brief period of time (on the order of 200 ms). Learn what isotopes are, how they differ from elements, and how they are represented and classified. explore the examples, characteristics, and applications of isotopes of hydrogen, carbon, chlorine, and nitrogen.

Isotope Diagram Carbon 12 is a stable isotope, while carbon 14 is a radioactive isotope (radioisotope). other than this, many synthetic carbon isotopes are synthesized in a lab environment and can only be sustained for a very brief period of time (on the order of 200 ms). Learn what isotopes are, how they differ from elements, and how they are represented and classified. explore the examples, characteristics, and applications of isotopes of hydrogen, carbon, chlorine, and nitrogen. For example, the naturally occurring isotope carbon 14 present in water is used to understand the age of water and other organic materials. read the iaea bulletins to learn more about the importance and uses of isotopes and their signatures. An isotope is an atom that has the same number of protons and a different number of neutrons. the atom is still considered the same element (still contains the same number of protons) as before but will have a different mass. Learn what isotopes are, how they are formed, and how they differ in properties and applications. find out how to notate, identify, and use isotopes for various purposes. Isotopes, isobars, isotones & isodiaphers definitions, examples, in chemistry: definition, types and importance of isotopes, isobars, isotones & isodiaphers.

Digital Illustration 3d Spheres With Electrons Neutrons And Protons For example, the naturally occurring isotope carbon 14 present in water is used to understand the age of water and other organic materials. read the iaea bulletins to learn more about the importance and uses of isotopes and their signatures. An isotope is an atom that has the same number of protons and a different number of neutrons. the atom is still considered the same element (still contains the same number of protons) as before but will have a different mass. Learn what isotopes are, how they are formed, and how they differ in properties and applications. find out how to notate, identify, and use isotopes for various purposes. Isotopes, isobars, isotones & isodiaphers definitions, examples, in chemistry: definition, types and importance of isotopes, isobars, isotones & isodiaphers.