
Difference Between Lanthanide Contraction And Actinide Contraction The chemical elements in the actinide series are called actinides, and the elements in the lanthanide series are called lanthanides. the main difference between actinides and lanthanides is that actinides can form complexes easily whereas lanthanides do not form complexes easily. Lanthanides and actinides are two prominent families of the periodic table of elements. discussed below are the differences between the elements in these two families and their common properties.
Lanthanide And Actinide Chemistry Ppt The main differences between lanthanides and actinides lie in their atomic numbers, periods in the periodic table, electronic configurations, chemical properties, and applications. While lanthanides help in understanding the electronic structure and properties of metals, actinides provide insights into the behavior of heavy elements, their radioactive decay, and their potential applications in various fields. Lanthanides, also known as rare earth elements, occupy the sixth period of the periodic table, following lanthanum. actinides, on the other hand, reside in the seventh period, following actinium. the lanthanides are generally considered less radioactive and more readily available than the actinides. Lanthanides on the other hand are commercially used for car parts, superconductors and magnets. actinides are highly radioactive and have increased propensity to undergo complex reactions. in contrast, lanthanides are have stable electronic configuration and do not readily undergo complex reactions.

Lanthanide And Actinide Chemistry Ppt Lanthanides, also known as rare earth elements, occupy the sixth period of the periodic table, following lanthanum. actinides, on the other hand, reside in the seventh period, following actinium. the lanthanides are generally considered less radioactive and more readily available than the actinides. Lanthanides on the other hand are commercially used for car parts, superconductors and magnets. actinides are highly radioactive and have increased propensity to undergo complex reactions. in contrast, lanthanides are have stable electronic configuration and do not readily undergo complex reactions. The primary distinction between actinides and lanthanides is that actinides may quickly form complexes, whereas lanthanides cannot. actinides are more radioactive, and lanthanides are not. Lanthanides are the steady, luminous cousins that add colour and strength to our world, while actinides are the dramatic relatives that power electricity and keep medicine moving forward. What are the main differences between lanthanides and actinides? the main differences between lanthanides and actinides lie in their electronic configuration, chemical behavior, and radioactivity. Lanthanides and actinides are two separate series of metallic elements found at the bottom of the periodic table. they are collectively known as inner transition elements because their differentiating electron enters the (n 2)f orbital, which is an inner orbital compared to the outermost shell.
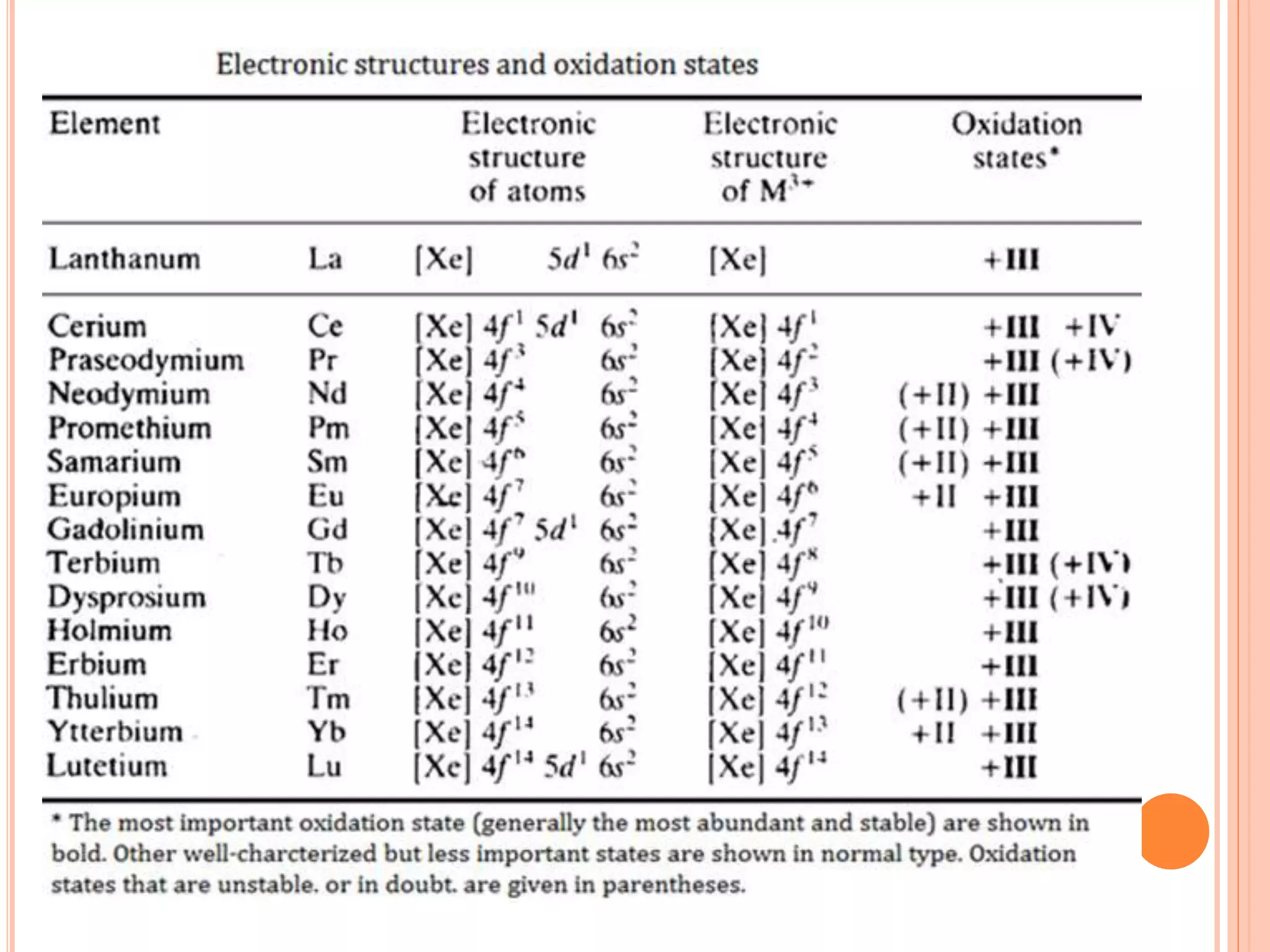
Lanthanide And Actinide Chemistry Ppt The primary distinction between actinides and lanthanides is that actinides may quickly form complexes, whereas lanthanides cannot. actinides are more radioactive, and lanthanides are not. Lanthanides are the steady, luminous cousins that add colour and strength to our world, while actinides are the dramatic relatives that power electricity and keep medicine moving forward. What are the main differences between lanthanides and actinides? the main differences between lanthanides and actinides lie in their electronic configuration, chemical behavior, and radioactivity. Lanthanides and actinides are two separate series of metallic elements found at the bottom of the periodic table. they are collectively known as inner transition elements because their differentiating electron enters the (n 2)f orbital, which is an inner orbital compared to the outermost shell.