Complex Ion Shape Exam Corner For each reaction, draw a diagram to illustrate the structure of, the shape of and the charge on the complex ion formed. Give the oxidation state of palladium in this compound. give the names of two possible shapes for the complex palladium ion in this compound.
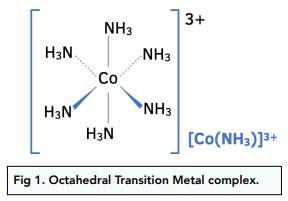
Complex Ion Shape Exam Corner This page describes the shapes of some common complex metal ions. it goes on to look at some simple examples of stereoisomerism (geometric and optical) in complex ions. Explore complex ions with interactive practice questions. get instant answer verification, watch video solutions, and gain a deeper understanding of this essential general chemistry topic. The shape of a complex is determined by the co ordination number and type of ligands bonded to a central metal atom or ion. the most common shapes are octahedral, tetrahedral, square planar and linear. Haemoglobin contains an iron(ii) ion bonded to five nitrogen atoms and one other ligand. the fifth nitrogen atom and the additional ligand are not shown in this diagram.
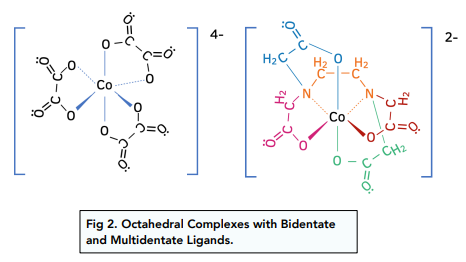
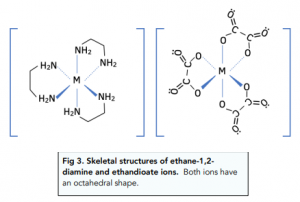
Complex Ion Shape Exam Corner The shape of a complex is determined by the co ordination number and type of ligands bonded to a central metal atom or ion. the most common shapes are octahedral, tetrahedral, square planar and linear. Haemoglobin contains an iron(ii) ion bonded to five nitrogen atoms and one other ligand. the fifth nitrogen atom and the additional ligand are not shown in this diagram. This page describes the shapes of some common complex metal ions. it goes on to look at some simple examples of stereoisomerism (geometric and optical) in complex ions. Transition metal ions commonly form octahedral complexes with small ligands (eg h2o and nh3 ). octahedral complexes can display cis–trans isomerism (a special case of e–z isomerism) with monodentate ligands and optical isomerism with bidentate ligands. (f) the compound [cr(nh3)4cl2]cl contains a complex ion that shows isomerism. draw the two isomers of the complex ion. state the type of isomerism shown. isomer 1. Your online learning platform for a levels, gce & grades 1 12. curated notes & resources. interactive quizzes & feedback. full exams & instant results. powerpoint learning materials. forgot password?.
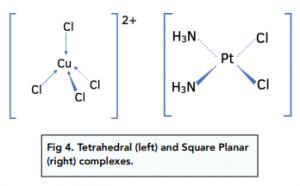
Complex Ion Shape Exam Corner This page describes the shapes of some common complex metal ions. it goes on to look at some simple examples of stereoisomerism (geometric and optical) in complex ions. Transition metal ions commonly form octahedral complexes with small ligands (eg h2o and nh3 ). octahedral complexes can display cis–trans isomerism (a special case of e–z isomerism) with monodentate ligands and optical isomerism with bidentate ligands. (f) the compound [cr(nh3)4cl2]cl contains a complex ion that shows isomerism. draw the two isomers of the complex ion. state the type of isomerism shown. isomer 1. Your online learning platform for a levels, gce & grades 1 12. curated notes & resources. interactive quizzes & feedback. full exams & instant results. powerpoint learning materials. forgot password?.
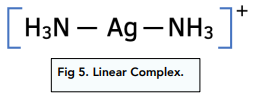
Complex Ion Shape Exam Corner (f) the compound [cr(nh3)4cl2]cl contains a complex ion that shows isomerism. draw the two isomers of the complex ion. state the type of isomerism shown. isomer 1. Your online learning platform for a levels, gce & grades 1 12. curated notes & resources. interactive quizzes & feedback. full exams & instant results. powerpoint learning materials. forgot password?.
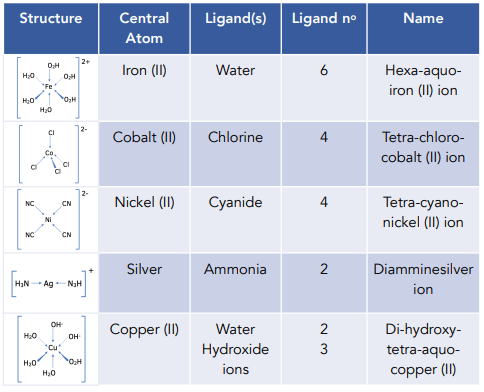
Introduction To Complex Ions Exam Corner