
Complex Ion Formation Pdf Coordination Complex Ion A complex ion forms from a metal ion and a ligand because of a lewis acid–base interaction. the positively charged metal ion acts as a lewis acid, and the ligand, with one or more lone pairs of electrons, acts as a lewis base. Many slightly soluble ionic solids dissolve when the concentration of the metal ion in solution is decreased through the formation of complex (polyatomic) ions in a lewis acid base reaction.
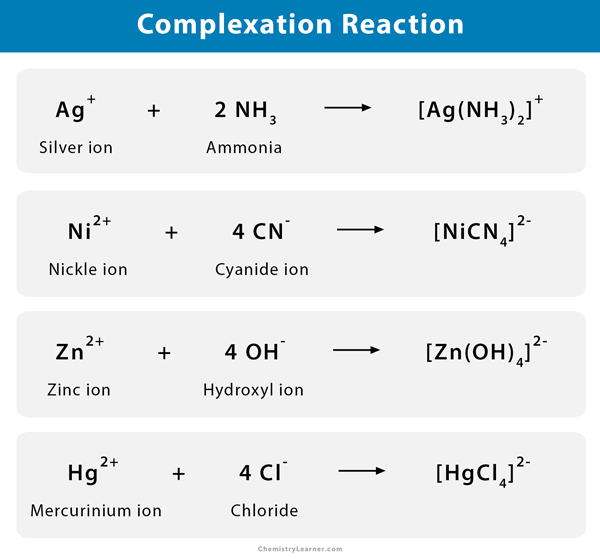
Complex Ion Formation Reaction Complex ion formation is a fundamental concept in chemistry, particularly within coordination chemistry. it involves the combination of metal ions with ligands to form stable, charged structures known as complex ions. Learn what a complex ion is, how it forms by co ordinate bonds with ligands, and how to name and classify it. see examples of complex ions with different metal ions and ligands, and how their coordination numbers vary. In this chemistry article, we learn about complex ions and their ligands, including the various types, the coordination number, and ligand substitution reactions. The formation of complex ions involves intricate interactions that depend on various factors, including the nature of the metal ion, the types of ligands involved, and the surrounding conditions.

Complex Ion Formation Reaction In this chemistry article, we learn about complex ions and their ligands, including the various types, the coordination number, and ligand substitution reactions. The formation of complex ions involves intricate interactions that depend on various factors, including the nature of the metal ion, the types of ligands involved, and the surrounding conditions. A hydrated ion is one kind of a complex ion (or, simply, complex), a species formed between a central metal ion and one or more surrounding ligands, molecules or ions that contain at least one lone pair of electrons, such as the [al (h 2 o) 6] 3 ion. Dive into the world of complex ions, exploring their definition, formation, and significance in inorganic chemistry laboratory settings. A complex ion, also known as a coordination complex, consists of a central metal atom or ion bonded to surrounding molecules or ions called ligands. these ligands donate electron pairs to the metal center, forming coordinate covalent bonds. To summarize, a complex ion is an adduct formed by the combination of a metal cation and a specific number of ligands, and it is crucial to recognize its representation in chemical formulas, particularly by the brackets that denote its structure.