
Actinides Vs Lanthanides What S The Difference Lanthanides and actinides are two prominent families of the periodic table of elements. discussed below are the differences between the elements in these two families and their common properties. Lanthanides are used in optical devices (night vision goggles), petroleum refining, and alloys. actinides are found primarily in applications where their radioactivity can be used to power devices such as cardiac pacemakers.

Lanthanides Vs Actinides Difference And Comparison The chemical elements in the actinide series are called actinides, and the elements in the lanthanide series are called lanthanides. the main difference between actinides and lanthanides is that actinides can form complexes easily whereas lanthanides do not form complexes easily. Lanthanides vs actinides understanding the definition of lanthanides and actinides by solving questions using real time examples and facts. Lanthanides, also known as rare earth elements, occupy the sixth period of the periodic table, following lanthanum. actinides, on the other hand, reside in the seventh period, following actinium. the lanthanides are generally considered less radioactive and more readily available than the actinides. The two rows that are generally placed underneath the main periodic table are called the lanthanide series and the actinide series. these two rows are produced when electrons are being added to f orbitals.

Lanthanides Vs Actinides Learn Definition Facts Examples Lanthanides, also known as rare earth elements, occupy the sixth period of the periodic table, following lanthanum. actinides, on the other hand, reside in the seventh period, following actinium. the lanthanides are generally considered less radioactive and more readily available than the actinides. The two rows that are generally placed underneath the main periodic table are called the lanthanide series and the actinide series. these two rows are produced when electrons are being added to f orbitals. Actinides are highly radioactive and have increased propensity to undergo complex reactions. in contrast, lanthanides are have stable electronic configuration and do not readily undergo complex reactions. Lanthanides and actinides differ from other elements in the periodic table due to their unique electronic configuration. they exhibit different chemical properties compared to the transition metals and the main group elements. Lanthanides are the steady, luminous cousins that add colour and strength to our world, while actinides are the dramatic relatives that power electricity and keep medicine moving forward. There is a distinct breakdown between gd and tb (lanthanide series) and between cm and bk (actinide series), which can be attributed to the small change in ionic radians made by the half filling of 4f and 5f shells respectively.
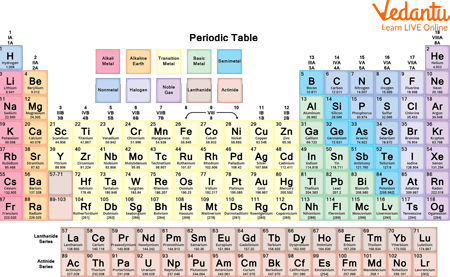
Lanthanides Vs Actinides Learn Definition Facts Examples Actinides are highly radioactive and have increased propensity to undergo complex reactions. in contrast, lanthanides are have stable electronic configuration and do not readily undergo complex reactions. Lanthanides and actinides differ from other elements in the periodic table due to their unique electronic configuration. they exhibit different chemical properties compared to the transition metals and the main group elements. Lanthanides are the steady, luminous cousins that add colour and strength to our world, while actinides are the dramatic relatives that power electricity and keep medicine moving forward. There is a distinct breakdown between gd and tb (lanthanide series) and between cm and bk (actinide series), which can be attributed to the small change in ionic radians made by the half filling of 4f and 5f shells respectively.

Lanthanides Vs Actinides Learn Definition Facts Examples Lanthanides are the steady, luminous cousins that add colour and strength to our world, while actinides are the dramatic relatives that power electricity and keep medicine moving forward. There is a distinct breakdown between gd and tb (lanthanide series) and between cm and bk (actinide series), which can be attributed to the small change in ionic radians made by the half filling of 4f and 5f shells respectively.