
Why Lanthanides And Actinides Are Separate On The Periodic Table Orango Study with quizlet and memorize flashcards containing terms like h, he, li and more. In the lanthanides, the contraction is about 4.4 pm per element; in the actinides, it is about 3 pm per element. the contraction is larger in the lanthanides than in the actinides due to the greater localization of the 4f wave function as compared to the 5f wave function.

3 The Periodic Table Except The Lanthanides And Actinides That Shows 3 the periodic table (except the lanthanides and actinides) that shows the transition metals capable of displaying cathodic and anodic electrochromism. Study with quizlet and memorize flashcards containing terms like hydrogen, helium, lithium and more. Study with quizlet and memorize flashcards containing terms like hydrogen, helium, lithium and more. Study with quizlet and memorize flashcards containing terms like h, he, li and more.

3 The Periodic Table Except The Lanthanides And Actinides That Shows Study with quizlet and memorize flashcards containing terms like hydrogen, helium, lithium and more. Study with quizlet and memorize flashcards containing terms like h, he, li and more. Click on this link for an interactive periodic table, which you can use to explore the properties of the elements (includes podcasts and videos of each element). State the periodic law and explain the organization of elements in the periodic table. predict the general properties of elements based on their location within the periodic table. The discovery of the periodic recurrence of similar properties among the elements led to the formulation of the periodic table, in which the elements are arranged in order of increasing atomic number in rows known as periods and columns known as groups. Periods are horizontal rows of the periodic table. they represent elements having the same number of electron shells or energy levels. however, each element has one more proton than its preceding element. thus, the periods are arranged according to the increasing atomic number of the elements.
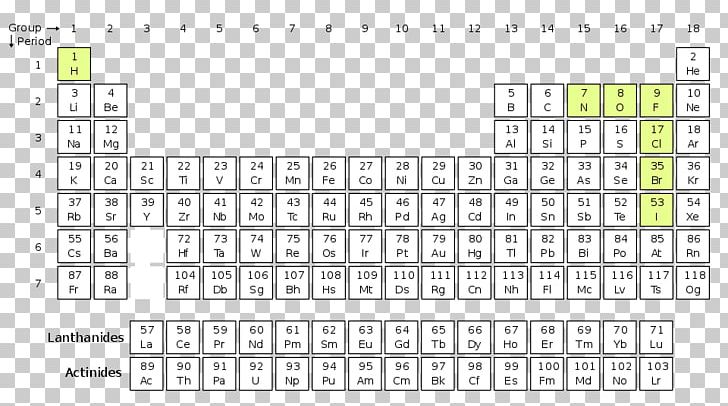
Lanthanides And Actinides Periodic Table Lanthanides And Actinides Click on this link for an interactive periodic table, which you can use to explore the properties of the elements (includes podcasts and videos of each element). State the periodic law and explain the organization of elements in the periodic table. predict the general properties of elements based on their location within the periodic table. The discovery of the periodic recurrence of similar properties among the elements led to the formulation of the periodic table, in which the elements are arranged in order of increasing atomic number in rows known as periods and columns known as groups. Periods are horizontal rows of the periodic table. they represent elements having the same number of electron shells or energy levels. however, each element has one more proton than its preceding element. thus, the periods are arranged according to the increasing atomic number of the elements.

Lanthanides And Actinides Periodic Table Actinides Mendeleev Table The discovery of the periodic recurrence of similar properties among the elements led to the formulation of the periodic table, in which the elements are arranged in order of increasing atomic number in rows known as periods and columns known as groups. Periods are horizontal rows of the periodic table. they represent elements having the same number of electron shells or energy levels. however, each element has one more proton than its preceding element. thus, the periods are arranged according to the increasing atomic number of the elements.
/141849999-56a12e8b3df78cf77268332b.jpg)
Why Lanthanides And Actinides Are Separate On The Periodic Table